BEST CASH PRICES on Viagra, Levitra, Cialis + FREE SHIPPING ... JUST TEXT 855.816.PALS ... Ready to order? Have Your Doctor Fax Your Prescription to 855.746.PALS. Dismiss
by Felicia Eddings, Pharm.D. | Apr 3, 2026 | News
Update [3/29/19] FDA is alerting health care professionals and patients of McDaniel Life-Line’s voluntarily recall of all lots of Life-Line Water. This product is being recalled because FDA analysis found the product to be contaminated with Pseudomonas aeruginosa....by Felicia Eddings, Pharm.D. | Apr 3, 2026 | News
[2/1/2019] FDA is warning compounders not to use glutathione-L-reduced powder distributed by Letco Medical, Decatur, Alabama, to compound sterile injectable drugs for patients. Based on reported adverse events, the agency has concerns with use of the Letco distributed...by Felicia Eddings, Pharm.D. | Apr 3, 2026 | News
Disclaimer: Early release articles are not considered as final versions. Any changes will be reflected in the online version in the month the article is officially released. Author affiliation: National Institute for Communicable Diseases, a Division of the National...by Felicia Eddings, Pharm.D. | Apr 3, 2026 | News
[12/11/2018] The U.S. Food and Drug Administration is alerting consumers, particularly males, not to purchase or use two e-liquids sold by HelloCig Electronic Technology Co. Ltd. that contain undeclared prescription drugs tadalafil and/or sildenafil. FDA laboratory...by Felicia Eddings, Pharm.D. | Apr 3, 2026 | News
Compounded drugs were produced under poor sterile production practices in an inadequately designed processingroom [12/7/2018] The U.S. Food and Drug Administration is alerting health care professionals and patients not to use drug products intended to be sterile that...
Click To Verify Security & Validation

Transferred Your Prescription?
 If you have not already done so, please CLICK HERE to have your prescription transferred into the Pill Pals Pharmacy Network.
With Pill Pals, You Get The BEST CASH PRICES On ALL Meds!
If you have not already done so, please CLICK HERE to have your prescription transferred into the Pill Pals Pharmacy Network.
With Pill Pals, You Get The BEST CASH PRICES On ALL Meds!
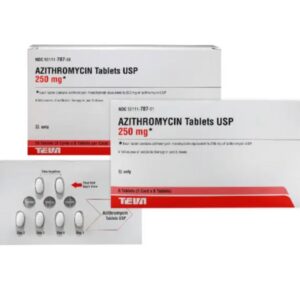 Azithromycin 250 mg Tablet Six (6) Pack --- TEVA Pharma --- Generic For Z Pack / Zithromax
Azithromycin 250 mg Tablet Six (6) Pack --- TEVA Pharma --- Generic For Z Pack / Zithromax
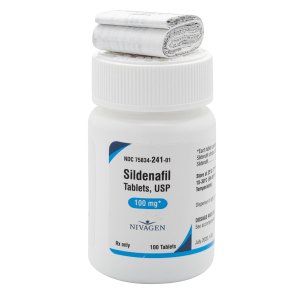 Sildenafil 100 mg Tablets --- Generic for Viagra --- Nivagen Pharma
$29.99 – $89.99Price range: $29.99 through $89.99 - or Auto-Reorder and Save 5%
Sildenafil 100 mg Tablets --- Generic for Viagra --- Nivagen Pharma
$29.99 – $89.99Price range: $29.99 through $89.99 - or Auto-Reorder and Save 5%
 Acetaminophen 500 MG Caplets ---1,000 Count --- Generic For Tylenol --- Major Pharma
Acetaminophen 500 MG Caplets ---1,000 Count --- Generic For Tylenol --- Major Pharma
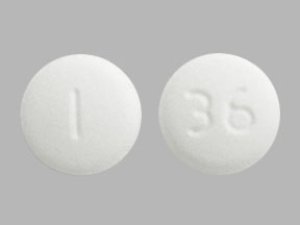 Sildenafil 50 mg Tablets --- Generic for Viagra --- Camber Pharma
$29.99 – $89.99Price range: $29.99 through $89.99 - or Auto-Reorder and Save 5%
Sildenafil 50 mg Tablets --- Generic for Viagra --- Camber Pharma
$29.99 – $89.99Price range: $29.99 through $89.99 - or Auto-Reorder and Save 5%
 Sildenafil 100 mg Tablets --- Generic for Viagra --- Torrent Pharma
$29.99 – $89.99Price range: $29.99 through $89.99 - or Auto-Reorder and Save 5%
Sildenafil 100 mg Tablets --- Generic for Viagra --- Torrent Pharma
$29.99 – $89.99Price range: $29.99 through $89.99 - or Auto-Reorder and Save 5%