BEST CASH PRICES on Viagra, Levitra, Cialis + FREE SHIPPING ... JUST TEXT 855.816.PALS ... Ready to order? Have Your Doctor Fax Your Prescription to 855.746.PALS. Dismiss
by Felicia Eddings, Pharm.D. | Mar 19, 2026 | News
Learn more about FDA’s resources to provide manufacturers with information on the review and approval process for biosimilar and interchangeable products.Guidance for IndustryFDA provides guidance on demonstrating biosimilarity and interchangeability, biosimilar...by Felicia Eddings, Pharm.D. | Mar 19, 2026 | News
Anuncio de seguridadInformación adicional para consumidoresInformación adicional para profesionales de la saludResumen de datosReferencias Anuncio de seguridad19 de septiembre del 2012. La Administración de Alimentos y Medicamentos de Estados Unidos (FDA por sus...by Felicia Eddings, Pharm.D. | Mar 19, 2026 | News
Anuncio de seguridadInformación adicional para consumidoresInformación adicional para profesionales de la saludResumen de datosFuentes Anuncio de seguridad13 de septiembre del 2012. La Administración de Alimentos y Medicamentos de Estados (FDA por sus siglas en...by Felicia Eddings, Pharm.D. | Mar 19, 2026 | News
[04-15-2011] To further evaluate the safety of Long-Acting Beta-Agonists (LABAs) when used in combination with inhaled corticosteroids for the treatment of asthma, the U.S. Food and Drug Administration (FDA) is requiring the manufacturers of LABAs to conduct five...by Felicia Eddings, Pharm.D. | Mar 19, 2026 | News
Safety AnnouncementTable 1. List of ondansetron products to be voluntarily withdrawn from the U.S. marketReference Safety Announcement[12-4-2012] The U.S. Food and Drug Administration (FDA) is notifying health care professionals that the 32 mg, single intravenous...
Click To Verify Security & Validation

Transferred Your Prescription?
 If you have not already done so, please CLICK HERE to have your prescription transferred into the Pill Pals Pharmacy Network.
With Pill Pals, You Get The BEST CASH PRICES On ALL Meds!
If you have not already done so, please CLICK HERE to have your prescription transferred into the Pill Pals Pharmacy Network.
With Pill Pals, You Get The BEST CASH PRICES On ALL Meds!
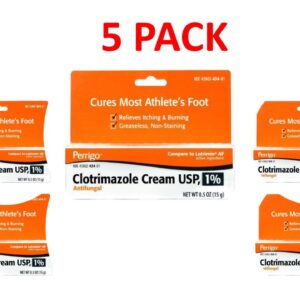 Clotrimazole 1 Percent (1%) Cream 15g (0.5 oz) --- Generic for Lotrimin AF Pack of 5 --- Perrigo Pharma
Clotrimazole 1 Percent (1%) Cream 15g (0.5 oz) --- Generic for Lotrimin AF Pack of 5 --- Perrigo Pharma
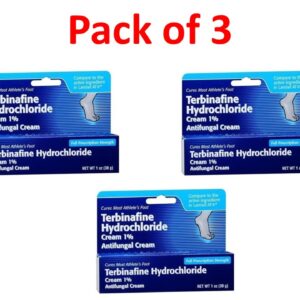 Terbinafine 1 Percent (1%) Cream 30 g (1 oz) --- Generic for Lamisil AT --- Pack of 3
Terbinafine 1 Percent (1%) Cream 30 g (1 oz) --- Generic for Lamisil AT --- Pack of 3
 Sildenafil 50 mg Tablets --- Generic for Viagra --- Amneal Pharma
$29.99 – $89.99Price range: $29.99 through $89.99 - or Auto-Reorder and Save 5%
Sildenafil 50 mg Tablets --- Generic for Viagra --- Amneal Pharma
$29.99 – $89.99Price range: $29.99 through $89.99 - or Auto-Reorder and Save 5%
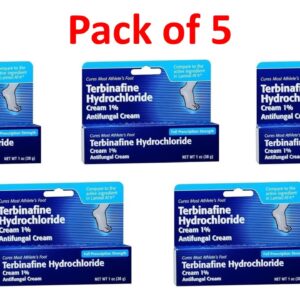 Terbinafine 1 Percent (1%) Cream 30 g (1 oz) --- Generic for Lamisil AT --- Pack of 5 --- Taro Pharma
Terbinafine 1 Percent (1%) Cream 30 g (1 oz) --- Generic for Lamisil AT --- Pack of 5 --- Taro Pharma
 Docusate Sodium /Senna 8.6-50 mg Tablets 100 Count --- Generic for Peri-Colace --- Rugby Pharma
Docusate Sodium /Senna 8.6-50 mg Tablets 100 Count --- Generic for Peri-Colace --- Rugby Pharma